Map by Dr. Richard Morrill, Emeritus Professor of Geography, University of Washington
Database architecture by Steve Hyde
by Dr. Dominic Corva, Executive Director
As the Washington State Legislature prepares to shut down medical marijuana access points by July 2016, it is helpful to begin to think about how many more I502 retail access points with medical cannabis endorsements will have to open to ensure the same level of geographic access Washington medical cannabis patients currently enjoy. Although available data is difficult to come by, this post is a first attempt at clarifying this picture for the public and policymakers.
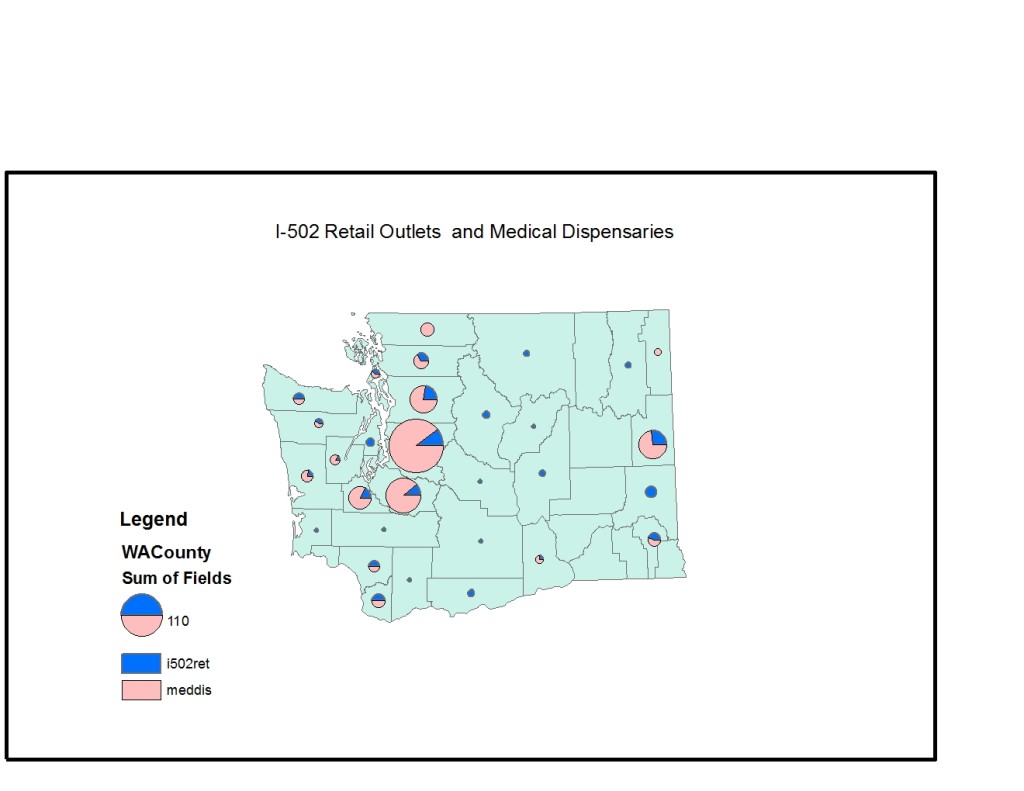
The map above represents county-level shares of approved (not necessarily currently open) I 502 retail points and DOR-identified registered, tax-paying medical cannabis businesses. There are many problems with this data. I 502 approved retail points are growing in number at a rate of approximately 3-4 a week: the rate of openings is unknown. On the medical side, we are not sure how many of the 450+ medical marijuana businesses registered with the Department of Revenue (DOR) are retail access points (brick and mortar or delivery) and how many might be taxpaying producer or processor businesses. The largest medical cannabis producer/processor-only business in the state, SODO-based New Leaf Enterprises (DAMA products), converted to I 502 status last month, so this number is also in motion.
“Patient access” is significant policy concern in the state of Washington, and the subject of much debate. Critics of the medical marijuana system, including I 502 business interests that have bulldozed the state’s longtime medical cannabis policy approaches in this legislative session for competitive rather than ethical reasons, have used the Seattle Times editorial board and anonymous “Staff” ( see here, here, and here, for starters) repeatedly over the last year to question the basic validity of the medical cannabis patient category. Medical marijuana advocacy organizations such as Americans for Safe Access [CASP’s fiscal sponsor is the federal 501(c)(3) sister organization, Americans for Safe Access Foundation – ed.] have spent a great deal of time and money pushing back against “cannabiz” strategies during this legislative session but with almost nothing to show for it.
So the question of access has been successfully framed as a question of the fundamental validity of cannabis as medicine, with zero efforts by anyone to collect real data on either patients or access. This post is agnostic on the question of “how many patients are real patients” because there is simply no data either on what constitutes authentic medical need, on the one hand, and what percentage of existing medical access point consumption is “valid.” Instead, we posit that absent real data about patients, we can instead provide real data about geographic access for however many existing and potential “real patients” are out there in the State.
Our ethical grounding point is the observation that if a single “authentic” patient loses access of any sort — geographical or socioeconomic — then bulldozing the medical cannabis landscape so that I 502 industry can gain competitive advantage is bad social policy. Medical cannabis policy originating from voter will through the 1998 initiative has had zero complaints from patients, wherever they are in the state, and instead has been accused of social harm by everyone but patients and doctors. Instead, accusations of social harm have demonstrably come from I 502 business interests that have lined the pockets of state lobbyists and legislators who have no knowledge of cannabis as herbal medicine; how cannabis products have evolved considerably from flower through the state’s medical cannabis system; and how this has been facilitated by the lack of punitive regulation introduced this legislative session.
As a result, the above landscape tells us something about ease of geographic access to (some kinds of) cannabis via the I 502 system for adult users, at the county level. The good news appears to be that in some rural counties, I 502 retail access points appear to provide access to cannabis for the first time (keep in mind our medical n only includes DOR-registered, taxpaying businesses). This is indicated by mostly blue circles.
But where the state’s population is concentrated, up and down the I-5 corridor and in Spokane to the East, and in some rural counties, patients will demonstrably lose access to retail medicine absent several changes over the next year and change.
First, I 502 stores are forbidden by law to include medical or therapeutic information through customer service or product packaging. This is substantially different from medical access points, most of whom provide some, admittedly sometimes accurate, information about therapeutic aspects of different varietals and “strains.” The paucity of accurate information in both systems is a significant policy concern, but it is not addressed by shutting down the system that actually provides some useful information, and legislating “medical endorsements” for some unknown number of I 502 retail points, some unknown number of which may be existing dispensaries that applied for but did not win favorable retail lottery placements in March 2014. In order for the state to provide as much geographical access for patients as is currently available according to DOR data, it will need to ensure that each of those circles represents the same number of medically-endorsed I 502 retail stores — some 430 across the state by July 2016.
Second, although cannabis testing standards and best laboratory practices in general are extremely problematic, the I 502 system has clearly exacerbated tendencies to reward labs that produce higher THC results. THC percentages are up 5-10% over previous ranges for medical markets. It’s quite clear that the I 502 system equates commercial “recreational” value with THC levels, reversing trends in medical markets to favor whole plant characteristics: varieties of cannabinoids especially CBD; and terpenes that may have as much or more therapeutic value than cannabinoid ratios. This is partially the result of stringent “security theater” regulations in I 502 that (a) keep the consumer from evaluating the product they are buying by smell and (b) severely limit visibility by requiring pre-packaging. Cannabinoid listing and “strain” name are pretty much the only things the I 502 consumer have to go on when it comes to purchasing flower. In order for the state to provide as much geographical access to diversity of flower terpenes and cannabinoids, as as is currently available according to DOR data, it will have to relax those packaging rules and police lab practices and standards set forth in the American Herbal Pharmacopeia that are currently being flouted widely in the interest of capturing lab market share. Recent statements by WSLCB indicate that they are aware of the problem and are taking promising steps.
There are many, many more problems with patient access that will be exacerbated by proposed changes to I 502 and medical cannabis policy, but this particular post is agnostic on these. Instead, it uses real data (with transparent limitations) to provide evidence that Washington state cannabis policy changes proposed in the legislature are bad social-geographic policy. The interests of Washington citizens — “society” at the state level — are poorly served by limiting geographic access to herbal medicine for patients, however many of them there are.